Photodynamic Inactivation: How Oxafence Dismantles a Virus
Traditional mask materials are designed to block and trap viruses. Oxafence introduces a different approach - one that inactivates viruses after they are captured.
At the core of this approach is a light-activated process known as antimicrobial photodynamic inactivation, which generates a highly reactive form of oxygen called singlet oxygen. Singlet oxygen has been shown in scientific literature to affect all three structural layers of a virus(1 2 3).
A Virus Is Only Infectious if Its Structure Is Intact
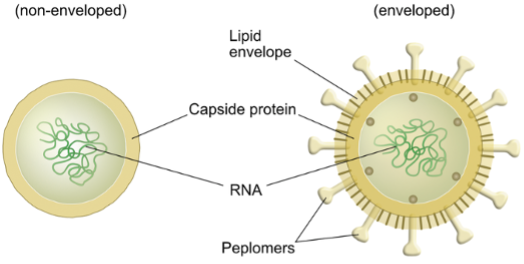
A virus is a microscopic infectious agent that must enter a living host cell to replicate. It remains infectious only as long as its structure is intact. A virus particle (virion) consists of:
Genetic material (RNA or DNA)
A protective protein shell (capsid)
In many cases, an outer lipid envelope
If any of these components are disrupted, the virus can lose its ability to infect. Singlet oxygen has been shown to interact with all three.(1 2 3)
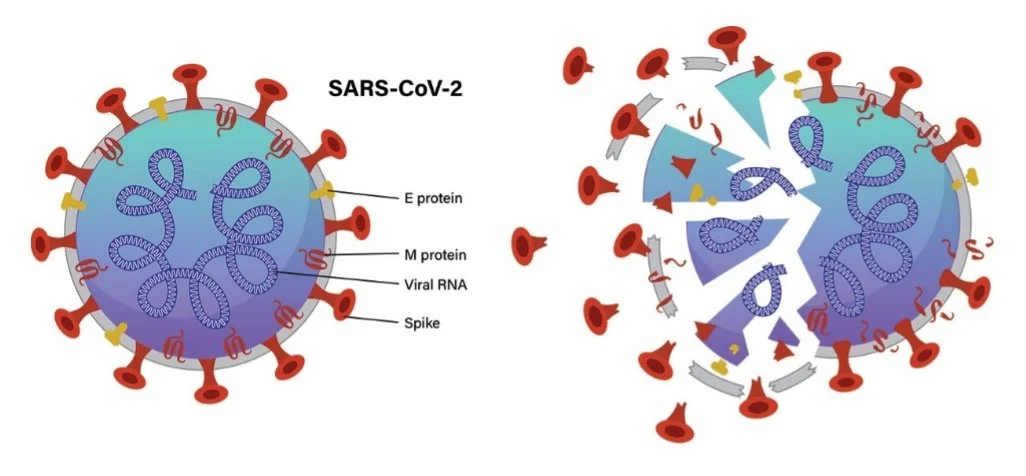
Step 1: Disrupting the Outer Envelope
Viruses such as SARS-CoV-2 and influenza rely on a lipid (fat-based) envelope to infect host cells. Singlet oxygen oxidizes this outer layer - gradually weakening its structure, much like how salt causes metal on a car to rust and weaken over time. As the envelope breaks down:
The virus loses its ability to bind to host cells
It becomes more vulnerable to additional damage (4 5)
Step 2: Disabling Critical Proteins
The protein shell (capsid) and surface proteins give the virus its structure and enable it to attach to host cells. Singlet oxygen can disrupt vulnerable regions of these proteins - like breaking a support pole in a tent, or dropping a phone in water where even a small amount of damage can cause the entire system to fail(6). Even small structural changes can prevent the virus from:
Recognizing host cells
Initiating infection
Step 3: Damaging the Genetic Material
At the core of the virus is its genetic material - the instructions for replication. Singlet oxygen can damage RNA or DNA strands, disrupting strands like ripping pages from a book, making it impossible for the virus to reproduce(7).
Scale, Speed, and Localization
Viruses are incredibly small – typically 100 to 250 nanometers, far smaller than a speck of dust – and singlet oxygen molecules are even smaller, at just 0.12 nanometers (roughly 700 to 1,000 times smaller than a virus like H1N1).(8)
A single photosensitizer molecule can generate 10,000 to 100,000 singlet oxygen molecules over its lifetime. On a theoretical surface area similar to that of a mask, more than a trillion singlet oxygen molecules can be generated every second when exposed to typical ambient indoor light.(2 5 9)
Each singlet oxygen molecule is short-lived – lasting less than 10 microseconds before returning to ordinary oxygen – and acts only in its immediate vicinity. But together, they form a relentless, invisible army. It’s like an endless wave of ants swarming a much larger intruder: each encounter may be small, but the cumulative effect can overwhelm and dismantle viral structures.(5 9)
Why This Mechanism is Broadly Effective
A virus becomes non-infectious if even one of its critical structural components is disrupted. Because singlet oxygen can affect:
The lipid envelope
Structural and surface proteins
Genetic material
It is considered a multi-target, broad-spectrum antiviral mechanism in the scientific literature.(1 2 3) While viruses have evolved different protective strategies, it is unlikely that all three structural layers can be fully protected simultaneously.(3 6 7)
Why This Matters on a Mask Surface
On a mask, pathogens can accumulate over time as they are captured from airflow. Traditional materials are designed to contain these particles. Oxafence introduces a mechanism to interact with them at the surface, helping address the accumulation of viable pathogens during real-world use.
Scientific Context
This overview describes the mechanism of singlet oxygen based on published research in photodynamic inactivation. It is intended to explain how Oxafence functions at a material level and does not describe or imply the performance of any specific product.
References
1. Tardivo JP et al. Photodiagnosis Photodyn Ther. 2005;2(3):175–191.
2. Hudson JB et al. Photochem Photobiol. 1997;65(5):723–729.
3. Wieding JU et al. Ann Hematol. 1993;67(6):259–266.
4. Córdoba-Lanús E et al. BMC Infect Dis. 2021;21(1):1169.
5. Lendvay TS et al. Infect Control Hosp Epidemiol. 2022;43(7):876–885.
6. Wigginton KR et al. Environ Sci Technol. 2012;46(3):12079–12087.
7. Ravanat JL et al. J Biol Chem. 2000;275(51):40601–40604.
8. Lobo CS et al. Photochem Photobiol Sci. 2022;21(6):1101–1109.
9. Dougherty TJ. Adv Photochem. 1992;17:275–311.